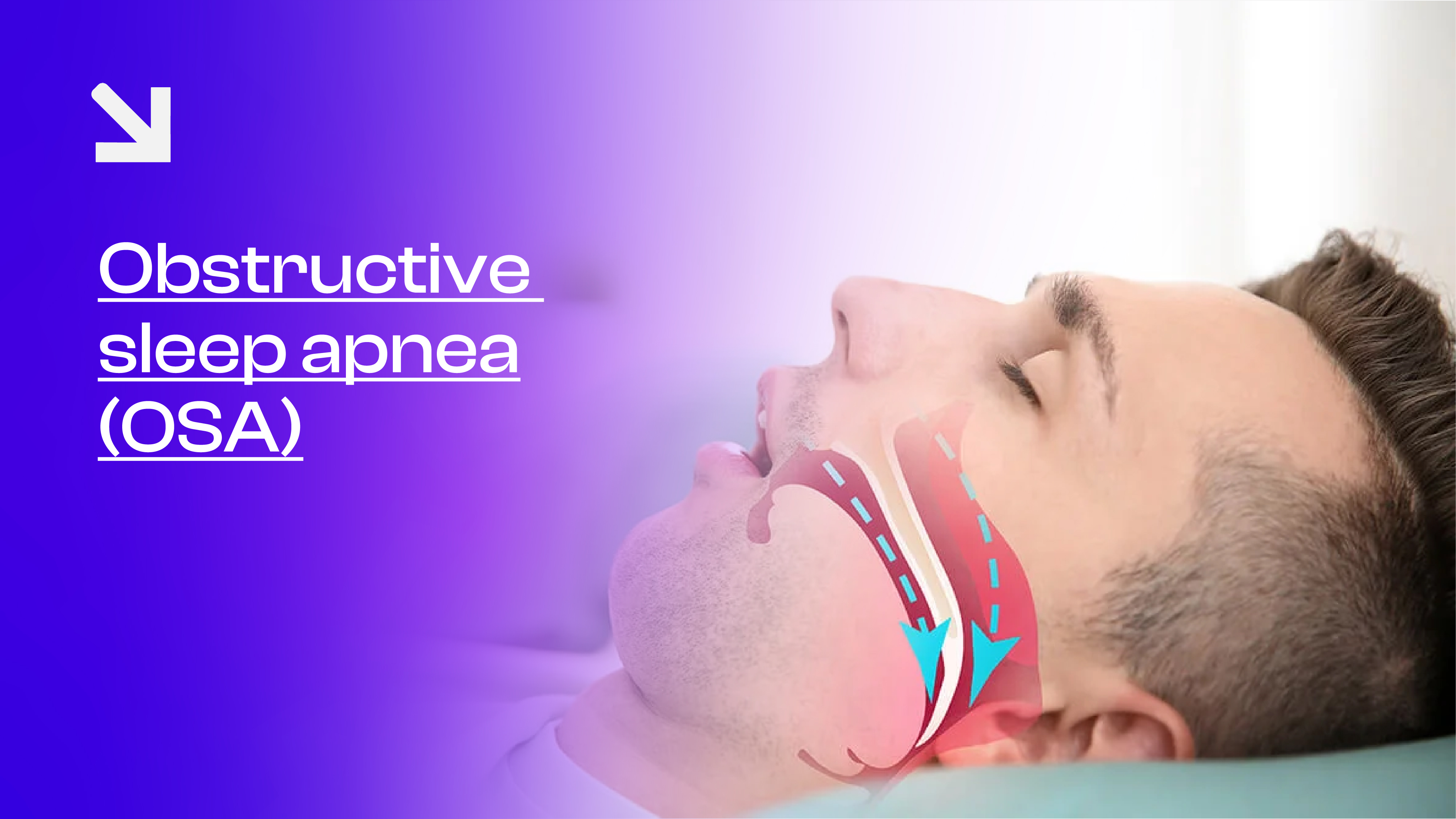
Overnight, obstructive sleep apnea (OSA) affects nearly one billion adults worldwide, many of whom do not find lasting relief with the gold-standard CPAP machine. Large masks, forced airflow, and noisy compressors contribute to non-adherence rates as high as 86% [1].
The iNAP device for sleep apnea, a mask-free negative air pressure device, introduces a new approach: intraoral negative-pressure therapy that is FDA-cleared and clinically validated. This in-depth overview explains the science, evidence, regulatory milestones, and why iNAP is emerging as a leading CPAP alternative device among modern sleep apnea treatment devices.
Struggling with CPAP? Explore the iNAP Device and request a live demo from Taevas Life Sciences today. https://taevaslifesciences.com/obstructive-sleep-apnea-device#get-in-touch-with-us

Introduction: A Paradigm Shift in Sleep Apnea Care
Conventional sleep apnea treatment has revolved around continuous positive airway pressure (CPAP), which pushes air into the airway to keep it open. However, wearing a mask nightly can be burdensome for many patients. Most individuals use CPAP for fewer than four hours per night, despite six to eight hours being required for optimal therapeutic benefit.
As a result, clinicians and engineers have sought alternatives to CPAP devices that maintain efficacy while improving comfort. One such innovation is the iNAP therapy for obstructive sleep apnea, an intermittent negative airway pressure device that applies controlled suction inside the mouth to draw soft tissues forward and maintain airway patency—without a mask or forced air.
This blog reviews the mechanism of action, key iNAP device clinical study findings, and its FDA 510(k) clearance status, positioning it as a promising new device for sleep apnea.
Mechanism of Action: A New Pathway to Patency
Unlike CPAP, which uses positive pressure to push the airway open, the iNAP device works through oral pressure therapy for OSA.
The system includes:
- A soft polymer mouthpiece (sometimes described as a tongue suction device for sleep apnea)
- A thin connecting tube
- A compact console
During sleep, the console generates regulated negative pressure (approximately −40 mmHg), gently drawing the tongue and soft palate forward to expand the retroglossal airway.
- Negative-Pressure Gradient: Uses vacuum-based stabilization instead of forced airflow.
- Intermittent Activation: Automatically regulates suction, conserving battery power (>8 hours per charge) and maintaining low noise (<30 dBA).
- Automatic Titration: Sensors continuously monitor and adjust intraoral pressure to maintain effectiveness.
By preserving natural nasal breathing and improving comfort, this pull-forward mechanism enhances adherence compared to many traditional sleep apnea devices.
Want to see the clinical data behind iNAP device efficacy? Explore published iNAP device clinical study results and understand how it compares to other CPAP alternative devices. https://taevaslifesciences.com/obstructive-sleep-apnea-device#get-in-touch-with-us

FDA Clearance: A Regulatory Milestone
On May 26, 2020, the iNAP One Sleep Therapy System by Somnics Health received FDA 510(k) clearance (K193460) under 21 CFR 872.5570 as an Intraoral Pressure Gradient Device.
- Classification: Class II medical device
- Indication: Adult OSA patients intolerant to positive airway pressure
- Predicate Device: Winx Sleep Therapy System (K130538)
- Bench testing confirmed consistent pressure delivery across varying temperature and humidity conditions.
- Biocompatibility testing met ISO 10993 standards.
- Clinical evidence demonstrated safety and performance consistent with FDA requirements.
This clearance establishes iNAP as an FDA-cleared OSA device and a regulated CPAP alternative device.
Clinical Validation: Cornerstone Studies
Multiple studies support the efficacy of this negative air pressure device.
- Hung et al. (2019)
Single-night crossover study (15 adults).
AHI reduction: 55.7% (23.1 → 9.7 events/h).
ODI reduction: 56.7%.
Seal adherence: 86.6% of the night [4]. - Cheng et al. (2021)
Multicenter study (N=32).
5% achieved ≥50% AHI reduction.
6 hours nightly use in 80% of participants [5].
- Nilius et al. (2024)
Long-term safety and efficacy cohort.
80% achieved AHI <10 within one month.
Average nightly treatment >6 hours [6]. - Camacho et al. (2016)
Meta-analysis of oral pressure therapy.
30–50% AHI reduction, comparable to low-pressure CPAP, with improved comfort [7].
These iNAP device clinical study findings demonstrate meaningful reductions in sleep-disordered breathing while improving real-world adherence.
Patient-Centered Outcomes: Beyond AHI
Clinical metrics are essential, but functional outcomes matter equally. Post-marketing findings indicate:
- 88% reported improved daytime alertness
- 90% maintained consistent nightly use (>6 hours)
- 73% average reduction in sleep-disordered breathing events
Such results reinforce iNAP’s role as an effective sleep apnea treatment device focused on comfort and usability.
iNAP vs. Alternatives to CPAP Devices
Several sleep apnea devices are available today.
A wide range of alternatives to CPAP devices has emerged [8], yet the iNAP device for sleep apnea stands out:
| Therapy Type | Mechanism | Invasiveness | Comfort | Portability |
| CPAP | Positive airflow | Non-invasive | Low (mask) | Moderate |
| MADs | Jaw advancement | Non-invasive | Moderate (TMJ risk) | High |
| EPAP | Exhalation valve | Non-invasive | Low (partial relief) | High |
| Inspire | Nerve stimulation | Surgical | High (no mask) | Low |
| iNAP | Negative oral pressure | Non-invasive | High | High |
The iNAP sleep apnea device balances efficacy, comfort, and ease of use through its mask-free interface, smart titration capability, and ultra-quiet performance.
Unlike a mandibular mouth device or a custom mandibular advancement device, iNAP does not reposition the jaw, reducing concerns about TMJ strain.
Patients researching treatment options often search for:
- Best mandibular advancement device
- Apnea mouth appliance
- FDA-approved sleep apnea mouth guard
- Mandibular advancement device cost
Within modern sleep technology products, iNAP represents a distinct, non-mandibular mechanism of action for treating obstructive sleep apnea.

Future Directions: Expanding Clinical Utility
Emerging developments include:
- AI-driven titration integration
- Expanded interface customization
- Ongoing research in broader sleep-related breathing disorders
- Reimbursement pathway expansion
These advancements strengthen iNAP’s position in the evolving OSA therapy device market.
Conclusion: A Clinically Validated CPAP Alternative
The iNAP device embodies a patient-centered approach to obstructive sleep apnea treatment. Supported by FDA clearance and robust iNAP device efficacy data, it provides a non-invasive, mask-free option among modern sleep apnea treatment devices.
For patients who struggle with CPAP adherence, iNAP offers a scientifically grounded and regulatory-approved alternative to CPAP devices.

Call to Action
If you or your patients are exploring a new device for sleep apnea, consider the iNAP device — a mask-free, FDA-cleared solution. Explore now.
Visit us to learn more, explore iNAP device reviews, inquire about iNAP device cost, or request a demonstration.
Experience clinically validated, comfortable iNAP sleep apnea treatment today.
References
- Lorenzi-Filho G, Almeida FR, Strollo PJ. Treating OSA: Current and emerging therapies beyond CPAP. Respirology. 2017;22(8):1500-1507.
- Somnics Inc. FDA Approves Somnics iNAP Sleep Therapy Pressure Titration Capability. Sleep World Magazine. Published June 23, 2023. https://sleepworldmagazine.com/2023/06/23/fda-approves-somnics-inap-sleep-therapy-pressure-titration-capability/. Accessed July 17, 2025.
- FDA. 510(k) Premarket Notification: K193460. Accessed July 17, 2025. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMN/pmn.cfm?ID=K193460.
- Hung TC et al. A novel intermittent negative air pressure device ameliorates obstructive sleep apnea syndrome in adults. Sleep Breath. 2019;23(3):849–856.
- Cheng CY et al. Evaluation of efficacy and safety of intraoral negative air pressure device in adults with obstructive sleep apnea in Taiwan. Sleep Med. 2021;81:163–168.
- Nilius G et al. Multicenter safety and efficacy study of a negative-pressure intraoral device in obstructive sleep apnea. Sleep Med. 2024;119:139–146.
- Camacho M et al. Oral pressure therapy (Winx) for obstructive sleep apnea: a meta-analysis. Sleep Breath. 2016;20(3):1011–1012.
- Harvard Health Publishing. Beyond CPAP: Other options for sleep apnea. Updated 2023. https://www.health.harvard.edu/staying-healthy/beyond-cpap-other-options-for-sleep-apnea. Accessed July 17, 2025.


